EEG and QEEG: Models and Theory
There are several models and theories that make an attempt at integrating the different findings or relating EEG patterns to treatment outcome. More specifically these are the ‘EEG Phenotype model’ initially developed and published by Jack Johnstone, Jay Gunkelman and Joy Lunt (Johnstone et al., 2005) and the ‘EEG Vigilance Model’ which was initially developed by Dieter Bente (Bente, 1964) and which is currently further investigated and developed by Ulrich Hegerl and his group (Hegerl et al., 2010; Hegerl, Olbrich, Schönknecht & Sander, 2008). In the following sections these models are explained in more detail focused on their application in ADHD and depression.
EEG Vigilance model
The regulation of vigilance and its flexible adaptation to internal and environmental needs are of fundamental importance for all higher organisms. Vigilance has to be adapted to the respective environmental situation, ensuring a high vigilance level in situations of danger and a reduced vigilance level during times of recreation. However, the interplay between environment and vigilance regulation also works the other way around: The environment actively created by a person can also depend on vigilance regulation. If the capacity of the brain to maintain a high vigilance level is reduced, a person will normally feel sleepy and thus seek an environment with low external stimulation and a chance to sleep. However, under certain circumstances such an unstable vigilance regulation can also induce a compensatory behavioral pattern termed here as ‘vigilance autostabilization behavior’. Hyperactivity, sensation seeking and other behavioral patterns create a highly stimulating environment. The resulting increase in external stimulation counteracts the impending vigilance decline and leads to a stabilization of vigilance. An everyday example would be the hyperactive, “high-spirited” behavior of overtired children. Related to this, mania has been described as sensation seeking gone out of control. By contrast, in times of a tonically high vigilance level, a person might avoid additional external stimulation and withdraw himself as autoregulatory behavior. The proposed concept of vigilance autostabilization behavior is related to earlier theories of brain function (Bente, 1964; Ulrich, Renfordt & Frick, 1986; Wundt, 1896), personality (Zuckerman, 1985) and sensation seeking (Eysenck, 1990).
EEG-vigilance algorithm “VIGALL”
In parallel to the transition from active wakefulness to deep sleep the human brain takes on different global functional states. These functional states are reflected in the spectral composition and topography of the EEG and have been termed vigilance stages. These states correspond to different levels of alertness at the behavioral level. Several stages can be separated during the transition from tense to relaxed wakefulness and further on to drowsiness until sleep onset.
In 1937, Loomis et al. (1937), later modified by Roth (1961), Bente (1964) and others (e.g. (Klimesch, 1999; Ulrich & Frick, 1986); proposed classifications for vigilance stages occurring during transition from active wakefulness to sleep onset. They are based on the following EEG phenomena during eyes closed, which have been demonstrated in several studies:
A1) Posterior alpha mostly seen after eye-closing with a frequency of 8-12 Hz and an occipital focus. This oscillation has been referred to as “idling rhythm” (Niedermeyer, 1997) because it marks a state of relaxed wakefulness, corresponding to vigilance stage A1 according to Bente (1964) and Loomis (Loomis et al., 1937).
A2-A3) Alpha power anteriorisation occurs increasingly after several minutes of relaxed wakefulness. Alpha peak frequency shows a slight decrease. This phenomenon is reported to occur during transition to drowsiness (Broughton & Hasan, 1995; Connemann et al., 2005; De Gennaro, Ferrara, Curcio & Cristiani, 2001; De Gennaro et al., 2004; De Gennaro et al., 2005; Pivik & Harman, 1995) and corresponds to vigilance stage A2 and A3 (Bente, 1964; Loomis et al., 1937).
B1) Low voltage EEG is increasingly observed during lower vigilance stages. The alpha rhythm disappears (alpha drop-out) and beta power increases (De Gennaro et al., 2001; Tanaka, Hayashi & Hori, 1996; Tanaka, Hayashi & Hori, 1997). This EEG pattern corresponds to vigilance stage B1 (Roth, 1961). The EEG in this state is similar to the EEG during intense mental activity and eyes open condition.
B2-3) Increased delta and theta activity is observed in parallel with increasing subjective drowsiness (Strijkstra, Beersma, Drayer, Halbesma & Daan, 2003; Tanaka et al., 1996; Tanaka et al., 1997), corresponding to vigilance stages B2 and B3 (Roth, 1961).
C) The occurrence of K-complexes and sleep spindles mark the beginning of definite sleep (Cash et al., 2009; De Gennaro & Ferrara, 2003; Tanaka et al., 1997).
Based on these EEG features a computer-based algorithm has been created for separating different EEG-vigilance stages (also see figure 4) for consecutive EEG segments. The first version of the algorithm “VIGALL” (Vigilance Algorithm Leipzig) was based upon the Fast Fourier-derived power of the four main EEG frequency bands alpha, beta, delta and theta during two-second segments of continuous EEG data at different sites. An improved second version of the algorithm now takes into account the intracortical source power (derived by Low Resolution Tomography-LORETA) of different regions of interest (ROIs).
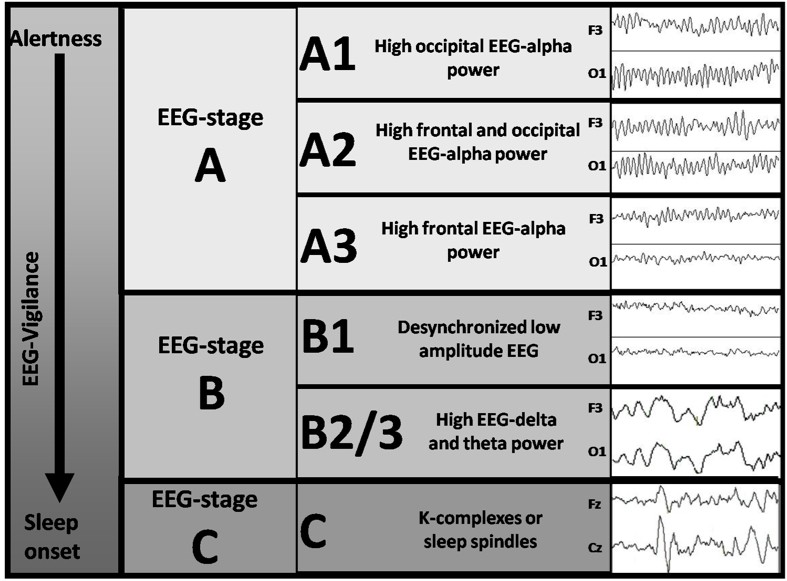 Figure 4: EEG-vigilance stages on the continuum from high to low vigilance levels (left column): The main criteria of the EEG-vigilance classification algorithm are given for six distinct EEG-vigilance stages (middle columns). Examples of typical two-second EEG curves are presented in the right column.
Figure 4: EEG-vigilance stages on the continuum from high to low vigilance levels (left column): The main criteria of the EEG-vigilance classification algorithm are given for six distinct EEG-vigilance stages (middle columns). Examples of typical two-second EEG curves are presented in the right column.
Several studies have further validated the EEG Vigilance model. Given the above stages and their relation to Vigilance, it is expected that switches between neighboring stages occur more often than switches to more distant stages, which was clearly demonstrated to be the case by (Olbrich et al., 2009) also see figure 5. This study demonstrated that switches between neighboring stages occurred significantly more often than a random process would reveal. These findings underline that the vigilance stage sequences during rest follow a certain order and give further validity to the EEG-vigilance algorithm VIGALL. Furthermore, Olbrich et al. (2009) also demonstrated that autonomic measures such as heart rate, also was lower for lower vigilance stages (e.g. B stages) as compared to higher vigilance stages (e.g. A stages).
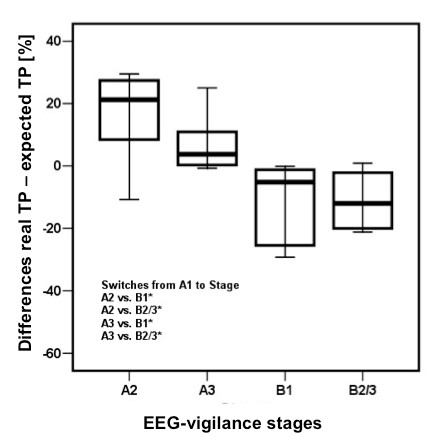
Figure 5: The differences between real (rTP) and expected (eTP) transition probabilities for stage A1 were significantly higher for switches to stages A2 and A3 than for switches to stages B1 and B2/3. This indicates that vigilance decline is a gradual process with switches between neighboring EEG-vigilance stages occurring more often than switches between distant vigilance stages.
EEG vigilance regulation in psychiatric disorders
As described earlier, changes in vigilance are also related to behavior. A decrease in vigilance or an ‘unstable’ vigilance regulation can lead to 2 different behaviors, 1) the organism decides to go to sleep and the vigilance reverts to sleep stages or, 2) the organism exhibits ‘autostabilization behavior’ to counter regulate their vigilance level such as hyperactivity and sensation seeking behavior. Figure 6 depicts this process in more detail. A physiological or ‘normal’ Vigilance regulation decreases over time. However, there are two deviating patterns of vigilance regulation – as can be seen below – namely the ‘rigid regulation’ and the ‘labile regulation’. The first example of rigid regulation is characterized by an inability to down-regulate one’s vigilance level and this person might avoid additional external stimulation and withdraw himself as autoregulatory behavior. This is a behavioral pattern, which is also often seen in depression. In contrast to this, individuals characterized by a labile regulation have an inability to maintain their vigilance level and/or exhibit unstable vigilance regulation. This type of vigilance regulation could induce a vigilance autostabilization behavior characterized by hyperactivity, sensation seeking and other behavioral patterns aimed at creating a highly stimulating environment. The resulting increase in external stimulation counteracts the impending vigilance decline and leads to a stabilization of vigilance. An everyday example would be the hyperactive, “high-spirited” behavior of overtired children. This behavioral pattern matches aspects of the behavior also seen in ADHD and mania.
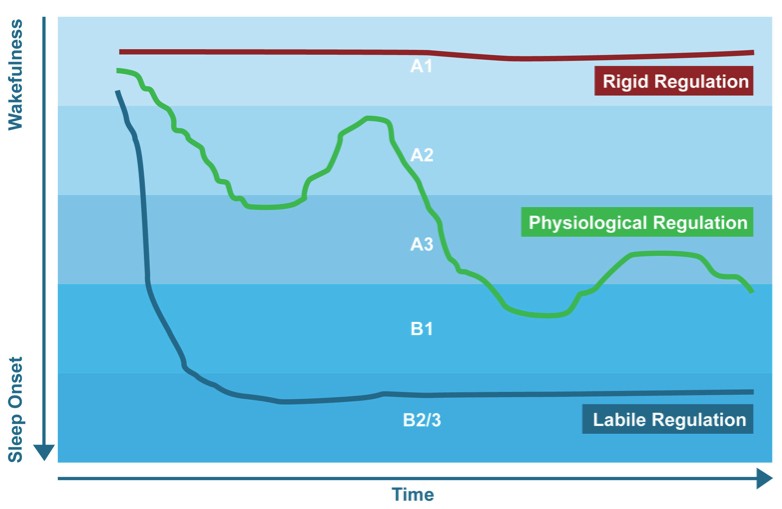
Figure 6: This figure shows the different modes of vigilance regulation, namely the rigid regulation, a physiological or ‘normal’ regulation and a labile vigilance regulation.
Mania and depression
Manic patients do not appear to be sleepy or tired. When evaluating EEG recordings of such patients, one would expect to find signs of a cortical hyperarousal. However, when studied under resting conditions with eyes closed, acutely manic patients consistently show rapid declines in vigilance within the first minute of EEG recording (Bschor, Müller-Oerlinghausen & Ulrich, 2001; Ulrich, Haug & Fähndrich, 1994; Van Sweden, 1986); a sub-group (19%) even shows signs of micro sleeps (defined as abrupt intrusion of sleep spindles) within the first 10 seconds into the EEG recording (Small, Milstein & Medlock, 1997). This finding generally has been neglected in theories on the pathophysiology of mania and is difficult to incorporate into current concepts. It does not appear to be a mere consequence of the sleep deficits often occurring within manic episodes. Instead, a causal role of the vigilance impairment in the pathomechanism of mania is suggested by the fact that sleep deficits can trigger or worsen hypomanic and/or manic syndromes in patients with bipolar disorders (BD) (Barbini, Bertelli, Colombo & Smeraldi, 1996; Hegerl et al., 2008; Wehr, 1992). Some symptoms of mania can be interpreted as autoregulatory reactions of the organism aimed to counteract the vigilance instability by increasing the level of external stimulation. While this might lead to vigilance stabilization, in many cases a vicious circle is initiated since this behavioral syndrome and the associated lack of sleepiness may aggravate the sleep deficit as well as the instability of vigilance regulation resulting in a vicious circle. According to this concept, most publications on treatment of mania using vigilance stabilizing agents such as Ritalin and modafinil reported an improvement within one or two hours of first dose (Beckmann & Heinemann, 1976; Brown & Mueller, 1979; Schoenknecht, Olbrich, Sander, Spindler & Hegerl, 2010).
In contrast to the unstable vigilance regulation in mania, a hyperstable vigilance regulation is observed during depressive episodes (Ulrich et al., 1994; Hegerl, Wilk, Olbrich, Schoenknecht & Sander 2011), in line with the earlier presented findings of increased alpha (thus reflective of high vigilance) in depression (Itil, 1983; Lemere, 1936; Pollock & Schneider, 1990). This goes in parallel with a difficulty falling asleep, an inner restlessness and a hyperactivity of the hypothalamic-pituitary-adrenal axis often found in depressed patients. One could hypothesize that depressive symptomatology with sensation avoidance and withdrawal may serve an autoregulatory function to counteract a hyperstable vigilance regulation. Also see figure 7 below for a case example of a bipolar patient recorded in his manic episode (top) and depressive episode (bottom) and the obtained EEG Vigilance stages. This example clearly demonstrates that – within subject – a labile vigilance regulation is associated with the manic phase, whereas a rigid vigilance regulation is associated with the depressive phase of the disorder.
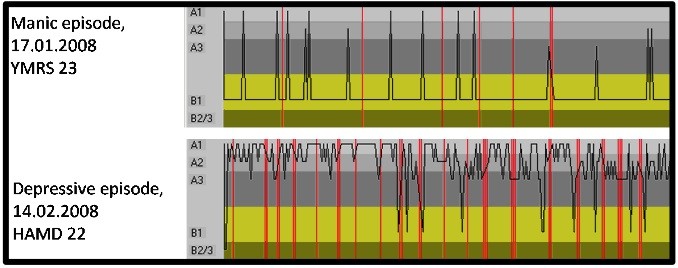
Figure 7: Time course of EEG-vigilance stages for consecutive two second segments of a ten minute resting EEG in a patient with bipolar affective disorder during a manic episode (top; Young Mania Rating Scale 23) and during a depressive episode (bottom; Hamilton depression Score 22). Labile vigilance regulation is found during the manic state while during the depressive state the vigilance level does not drop to low vigilance stages. Vertical red lines mark segments with artifacts.
Vigilance regulation in ADHD
Support for an unstable vigilance regulation in ADHD is provided by the fact that this disorder is associated with sleepiness, shortened sleep latency (Golan, Shahar, Ravid & Pillar, 2004), primary sleep disorders, sleep related movement disorders and parasomnias (Chervin et al., 2002; Konofal, Lecendreux & Cortese, 2010; Walters, Silvestri, Zucconi, Chandrashekariah & Konofal, 2008) and ADHD-like behavior can be induced in children by sleep restriction (Fallone, Acebo, Arnedt, Seifer & Carskadon, 2001; Golan et al., 2004). Recently, Van Veen (Van Veen et al., 2010) reported in a sample of adult ADHD patients that 78% had sleep-onset insomnia, confirmed by actigraphy and associated with a delayed nighttime melatonin onset. A similar rate of 73% sleep onset insomnia has been reported in children with ADHD (Van der Heijden, Smits, Van Someren & Gunning, 2005) and normalizing this sleep onset insomnia by for example melatonin or chronotherapy results in clinically meaningful improvements in ADHD/ADD symptomatology (Dahl, Pelham & Wierson, 1991; Hoebert et al., 2009).
Taken together, these data – along with the earlier reported EEG findings found in ADHD – suggest that a labile vigilance regulation is a pathogenic factor in ADHD. Some symptoms of ADHD can be seen as a direct result of the unstable vigilance regulation (deficits in sustained attention, distractibility), while other symptoms (e.g. hyperactivity, “sensation seeking”) can be interpreted as vigilance stabilizing syndrome, as is summarized in figure 8 below. Therefore, the well-documented effectiveness of psychostimulants in pediatric ADHD (Faraone & Buitelaar, 2009; Pliszka, 2007) is most likely explained by its vigilance stabilizing property.
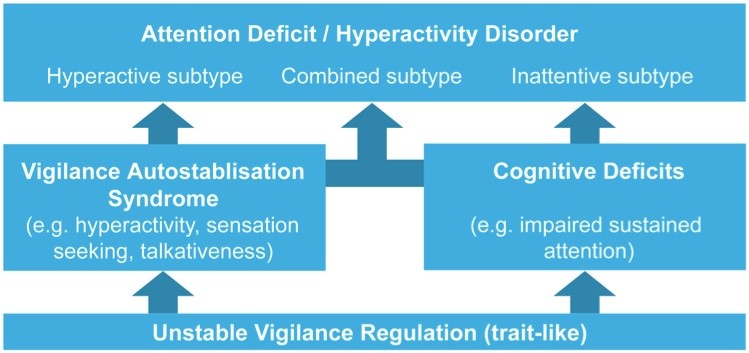
Figure 8: This figure provides an overview of the relation between an unstable vigilance regulation and the behavioral symptoms of ADHD (adapted from Hegerl et al. 2009).
EEG Phenotype model
As described earlier, the concept of endophenotypes was coined as early as in 1966 by John and Lewis (1966) and further developed based on studies in schizophrenia by Gottesman and Shields (1967; 1972). The idea behind an endophenotype is that it is the intermediary step between genotype and behavior and thus is more closely related to genotype than behavior is. Endophenotypes can be investigated to yield more information on the underlying genotype. In parallel there have also been many studies using the term biological marker, trait, biomarker etc. Here it is important that in line with Gotessman and Gould (2003), an ‘endophenotype’ refers to a marker when also certain heritability indicators are fulfilled, whereas a ‘Biomarker’ simply refers to differences between patient groups, which do not necessarily have a hereditary basis.
In 2005 Jay Gunkelman and associates submitted a paper proposing a set of EEG patterns as “EEG phenotypes” when the genetic links were known, and as “candidate EEG phenotypes” when the linkage to genetics remained unknown (Johnstone et al., 2005). These proposed EEG-based phenotypes are stable states of neurophysiological function, and can be identified from the raw EEG waveforms. The authors proposed a framework, which permitted researchers and clinicians to describe much of the observed EEG variance with a small number of categories of phenotypical divergence. These groupings are not identical to the DSM-IV groupings, and they are observed to cut across the DSM-IV categories. Unlike the DSM-IV, these phenotypes were observed to predict an individual’s response to both neurofeedback and medication approaches to therapy.
The literature on medication response prediction suggests that a phenotypic perspective may help enhance efficacy when prescribing medication, as seen in the work by Suffin & Emory (1995) who demonstrated that patients with frontal theta responded better to stimulant medication and patients with frontal alpha responded better to an antidepressant, irrespective of their DSM diagnosis. This method is referred to as referenced EEG or rEEG, and its efficacy has recently been replicated in a larger controlled study (Debattista et al., 2010). Furthermore, Prichep et al. (1993) found the same results in Obsessive Compulsive Disorder (OCD) where OCD patients with excess alpha responded better to an SSRI, as compared to OCD patients with excess theta. Improved outcomes may also be seen in neurofeedback, as demonstrated in the clinical outcome improvement reported by Wright and Gunkelman (1998) when they added the EEG phenotype approach to guide neurofeedback. Using a slightly different approach, Monastra, Monastra and George (2002) demonstrated that using a pre-selection on excess theta/beta ratio in an ADHD population also improved treatment outcome to neurofeedback (resulting in a doubling of the effect size) employing theta/beta neurofeedback, further supporting this notion.
Table 1: A summary of the EEG Phenotypes originally proposed by Johnstone, Gunkelman and Lunt (2005). The top part of the table represents EEG Phenotypes for which hereditability and/or genetic linkages have been reported (also summarized below) and the bottom part reflects ‘candidate EEG phenotypes’ for which more research is required to establish clear hereditability and/or genetic linkages.
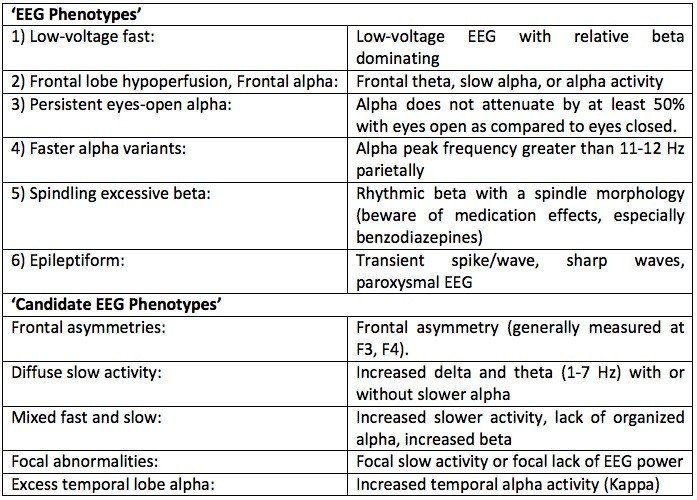
Many studies have investigated the heritability of the EEG in twin studies and family studies (see: Martinovi, Jovanovi, & Ristanovi, 1997; Vogel, 1970), and found that many aspects of the EEG are heritable. In a meta-analysis van Beijsterveld and van Baal (2002) demonstrated high heritability for measures such as the APF (81%), alpha EEG power (79%), P300 amplitude (60%) and P300 latency (51%), all suggesting that EEG and ERP parameters fulfill the definition of an endophenotype. Table 1 shows an overview of the original EEG Phenotypes proposed by Johnstone, Gunkelman and Lunt (2005). Below a summary is provided about what is currently known about the specific EEG Phenotypes as proposed by Johnstone, Gunkleman and Lunt (2005) and their underlying genetics and heritability:
- Low-voltage (alpha) EEG: This is the most well described EEG phenotype to date and was first described by Adrian and Matthews (Adrian & Matthews, 1934). The latter author exhibited an EEG in which alpha rhythm ‘…may not appear at all at the beginning of an examination, and seldom persists for long without intermission.’ (Adrian & Matthews, 1934: page 382). The low-voltage alpha EEG has been known to be heritable (autosomal dominant) and the heritability of alpha power is estimated at 79-93% (Anokhin et al., 1992; Smit et al., 2010; Smit, Posthuma, Boomsma, & De Geus, 2005; Vogel, 1970; Beijsterveld & van Baal., 2002). Low-voltage EEG is a well-described endophenotype in anxiety and alcoholism (Ehlers, Garcia-Andrade, Wall, Cloutier, & Phillips, 1999; Enoch, Schuckit, Johnson, & Goldman, 2003; Bierut et al., 2002; Enoch et al., 1999; Pine & Pine, 1953). Alpha power and LVA have been successfully associated with a few chromosome loci (Enoch et al., 2008) but also with single genes: a serotonin receptor gene (HTR3B) (Ducci et al., 2009), corticotrophin releasing binding hormone CRH-BP (Enoch, White, Waheed, & Goldman, 2008), a gamma-amino butyric acid (GABA)-B receptor gene (Winterer et al., 2003) and with the BDNF Val66Met polymorphism in depression (Veth, Arns, Drinkenburg, et al., in preparation).
- Frontal alpha: In addition to the high heritability of parieto-occipital alpha power referred to above, heritability of alpha at frontal sites is also high (85-87%) (Anokhin et al., 2006) but generally lower as compared to parieto-occipital sites (van Beijsterveldt & van Baal, 2002).
- Persistent eyes open alpha: Vogel (1970) also described a ‘Monotonous High Alpha Waves’ pattern, a characteristic that is heritable in a simple autosomal dominance manner. The description of this EEG pattern (‘Kontinuität’) is very similar to the ‘hyperrigid’ EEG described in the EEG Vigilance model.
- Faster alpha variants: The alpha peak frequency (APF) has been shown to be the most reproducible and heritable EEG aspect (Posthuma, Neale, Boomsma, & de Geus, 2001; Smit et al., 2005; van Beijsterveldt & van Baal, 2002) and has been associated with the COMT gene, with the Val/Val genotype being marked by a 1.4 Hz slower APF as compared to the Met/Met group (Bodenmann et al., 2009); this difference could explain a considerable amount of variability in this measure.
- Spindling excessive beta: Family studies have shown that frontal and fronto-central beta spindles and excess beta exhibit an autosomal dominant mode of inheritance in healthy persons, but these patterns can also occur as a result of brain damage. Furthermore, the pattern of fronto-precentral beta has a lower frequency in Japanese (Vogel, 1970). A strong linkage between beta frequencies and GABA-A receptor genes has been reported, in line with the often-reported medication effects of benzodiazepines resulting in a ‘beta buzz’ (Porjesz et al., 2002).
- Epileptiform EEG: Several types of paroxysmal EEG or epileptic EEG have also been demonstrated to be heritable and genetically linked (Haug et al., 2003; Kaneko, Iwasa, Okada & Hirose, 2002; Vaughn, Greenwood, Aylsworth & Tennison, 1996), however as mentioned before neurological EEG falls outside the scope of this thesis.
The EEG phenotype model demonstrates overlap with the EEG vigilance model, where in the EEG Phenotype model an EEG is described as its predominant feature (e.g. frontal alpha, frontal theta) the EEG vigilance model describes these as part of a continuum of vigilance. This thesis will investigate both models and aims to integrate findings from both models in their relation to predicting treatment outcome in ADHD and depression.
One critical point must be remembered when viewing the listing in table 1: the various phenotypes may coexist. The various combinations of the phenotypes are too numerous to be handled completely in this limited chapter presentation. Thus, this list should not be construed as a replacement for professional assistance in designing a neurofeedback intervention or in prescribing medication, nor in any way can this be used to fully characterize an individual’s EEG/QEEG.
